Biotech firm offering cosmetic companies an alternative to animal testing to open in US
Genoskin spun off from the French National Center for Scientific Research and the Paul Sabatier University in 2011. Last year the biotech venture became profitable and reports that in 2016 80% of revenue came from exports of its skin models and that a significant portion of those exports were to the US.
“The US market is of key importance to us,” affirms Pascal Descargues, founder and CEO of Genoskin, in a press release. “Genoskin already has customers in the region. US sales represented 30% of the company’s overall turnover in 2016.”
Today the company announced that Genoskin has formally expanded its business into the North American region, opening an office in Boston, Massachusetts. And, in 2018 Genoskin will open production facilities here in the US too.
Simulacrum
Safety and efficacy testing are an essential part of cosmetic and personal care product development. And many beauty makers as well as companies that support the industry have been working to find suitable alternative to animal testing and in vivo human testing.
“Animal testing is inefficient, time-consuming, expensive and increasingly perceived as unethical,” explains Pascal Descargues, founder and CEO of Genoskin, in a press release about the US expansion.
“We believe our technology marks a turning point,” says Descargues. “Not only do our human skin models address all these issues, they also help eliminate the major problem of efficacy and toxicity issues that arise due to differences between animal and human tissue.”
“Our skin models help academic institutions, pharma, cosmetic and chemical companies obtain more predictive results in order to lower R&D costs.”
Biotechnology
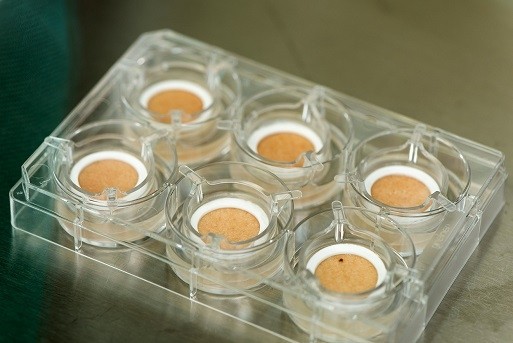
The Genotech human skin test models are covered by a US patent and, according to the company, “to date, there are no equivalent skin models on the US market.”
Genotech models aren’t produced, manufactured, or printed the way that some other alternative testing mediums are. The company essentially recycles skin that would be discarded after common surgeries (like tummy tucks). “The retrieved skin is placed in a testing well that contains a special biological matrix to keep the skin alive for several days,” as the company press release explains it.